Michael Gwarisa
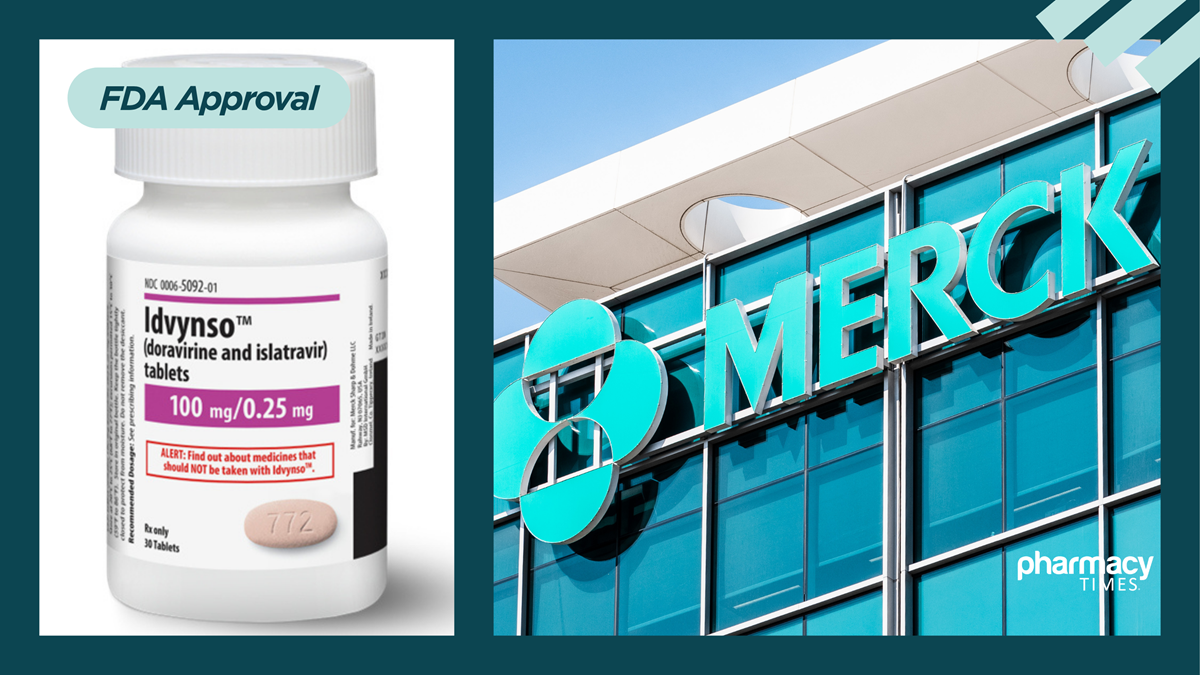
In a move that could significantly reduce pill burden and improve adherence to antiretroviral therapy (ART), the United States Food and Drug Administration has approved a new once-daily pill developed by Merck & Co. for adults living with HIV-1 who are already virologically suppressed. The pill is intended to replace a patient’s existing antiretroviral regimen.
The therapy, known as Idvynso, combines doravirine and islatravir into a single daily dose. Doravirine is already available as Pifeltro, while islatravir remains under investigation in several clinical trials exploring both daily and weekly HIV treatment and prevention options.
“IDVYNSO combines islatravir, a next-generation NRTI with multiple mechanisms of action, including translocation inhibition, with doravirine, an NNRTI with an established efficacy and safety profile. As the only two-drug, non-INSTI, tenofovir-free regimen, IDVYNSO expands therapeutic diversity beyond the currently available oral treatment options,” said Dr. Eliav Barr, senior vice president and chief medical officer, Merck Research Laboratories.
“As the health needs of adults living with HIV change over time, IDVYNSO gives clinicians a new choice for HIV treatment. This approval marks an important new chapter in Merck’s long-standing commitment to research and discovery for people living with HIV.”
Approval of the regimen came ahead of a key regulatory target date set under the Prescription Drug User Fee Act. This signals confidence in the drug’s safety and effectiveness based on available data. The combination previously met its primary goals in two Phase 3 clinical trials involving patients who had already achieved viral suppression. Additional studies have also shown promising outcomes in other treatment settings, strengthening its profile as a potential alternative to existing therapies.
A once-daily two-drug regimen represents an important shift in HIV care. Traditional ART often involves multiple medications taken daily, which can be challenging for long-term adherence. Simplified regimens are widely seen as a way to improve treatment consistency, reduce side effects, and enhance quality of life for people living with HIV.
For Merck, the approval marks a strategic step toward expanding its footprint in HIV treatment and prevention. The company is aiming to build a multibillion-dollar portfolio in this space, supported by a pipeline that includes long-acting therapies. Among these is a once-monthly pre-exposure prophylaxis pill currently in late-stage clinical development, which could further transform HIV prevention if successful.
Despite its long history in HIV research, Merck has in recent years focused more heavily on oncology, driven largely by the success of its cancer immunotherapy drug Keytruda. HIV has therefore represented a relatively small share of the company’s overall revenue and research investment.
Merck’s involvement in HIV dates back to 1986, during the early years of the global epidemic. A decade later, it introduced Crixivan, one of the first protease inhibitors, which received rapid regulatory approval at the time. That milestone helped shape the evolution of combination antiretroviral therapy, now the standard of care worldwide.
The HIV treatment market remains highly competitive. Major players include Gilead Sciences and ViiV Healthcare, the latter largely owned by GlaxoSmithKline with participation from Shionogi. Pfizer has also had a stake in ViiV, although it recently announced plans to exit.
HIV treatments and prevention medicines continue to be a major revenue driver for Gilead, accounting for a significant portion of its earnings. Like Merck, the company is also working on next-generation therapies aimed at improving convenience and adherence, including simplified daily regimens.
The introduction of a once-daily combination like Idvynso highlights ongoing efforts to make HIV treatment more manageable. As innovation continues, the focus is increasingly shifting toward reducing dosing frequency while maintaining strong viral suppression, a balance that could reshape long-term HIV care.