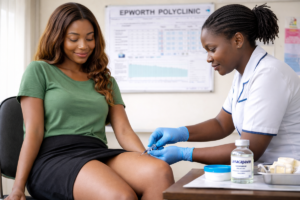
Professor Qin Zhiqing, an Associate Professor at the China National Institute of Parasitic Diseases (NIPR) has been in Zimbabwe for the past two months and together with a team from the National Institute for Health Research (NIHR), a department in the Ministry of Health and Child Care (MoHCC), they have been conducting a Schistosomiasis pilot study in Shamva District.
By Michael Gwarisa
As part of their study, they are also assessing the use of Rapid Diagnostic Testing (RDT) to detect and identify Schistosomiasis Cases in communities. At a laboratory site in Madziva, Professor Zhiqing is holding two test kits in both his hands. The other test kit is showing a positive reading for Schistosomiasis.
Haematobium (S.Haematobium) or Urinary Bilharzia. The sample has been collected from a Serum sample since the test kits at the lab currently are only testing for Intestinal Bilharzia or Schistsomsisi Mansonai. On the other hand, he is holding a control test kit which is meant to show that the kit is not defective in any way.
This is a breakthrough,” he says while holding the test kit with a positive reading from a Serum or blood sample.
“You can see the bands here, this one is a positive and shows the presence of S.Haematobium, a Urinary Bilharzia infection.”

Should the results from the ongoing intestinal Rapid Diagnostic test be successful, the team at the lab through support from their Chinese counterparts, will develop test kits for Urinary Schistosomiasis testing. This could revolutionize Zimbabwe’s Schistosmsis surveillance and screening at the community level and could see Zimbabwe soon moving away from relying solely on microscopy as the test kits will be rolled out countrywide since the disease is endemic in all provinces.
According to data from a recently conducted Geospatial Survey on Neglected Tropical Diseases (NTDs) in Zimbabwe, in 2021 Mashonaland Central and Mashonaland East Provinces led in terms of Schistosomiasis prevalence, with Masholand Central recording a prevalence of 19.0 percent, followed by Mashonaland East with a prevalence of 13.6 percent. Mash East is followed by Manicaland Province which has 11.8 percent, Masvingo 10.8 percent, Mash West with 9.0 and Midlands province with 7.4 percent. The rest of the provinces have a prevalence below 5 percent.
Currently, in Zimbabwe, the available diagnostic methods for schistosomiasis are those that rely on stool and urine microscopy for parasite detection. These include urine microscopy, serum antibodies, antigen detection, and the detection of the DNA of the parasite. However, the turnaround time for results under the blood samples or Serum route is longer compared to the Rapid Test kits. Microscopic diagnosis is happening at the District level. The Rapid Diagnostic kits will improve diagnostic at the community or village level.
Sharon Magudu, a Laboratory scientist working at Madziva Lab said the new test kits produces results in just 25 minutes.
“We are still developing these kits. If this one works just like you are seeing in all these positives, it means we have come to our breakthrough. We are still using these test kits to screen for intestinal Bilharzia using stool samples. Very soon, we are going to be moving to Urinary Bilharzia which is the Schistosoma haematobium, the one where one passes out urine with blood. We can use the urine to test on the rapid test. So it is going to be very fast, we just get the sample and after 25 minutes, it reads,” said Sharon.
However, as for the microscopy or Serum or blood samples, the process is a bit lengthy as it involves the collection of samples, lab tests, and viewing under a microscope. The process takes several hours for results to show.
Professor Nicholas Midzi, the Director National Institute for Health Research (NIHR) said Zimbabwe could soon move from serum or blood testing, to urine and intestinal or stool testing, using the Rapid Diagnostic Tools.
“There are other tools that are now being assessed. In the community, we need point-of-care diagnostic tools that then can be used at health facilities in the community without the need for major expertise. The Rapid Diagnostic Tool has been recently developed. We want to see if they can be able to detect urinary Bilharzia or intestinal Bilharzia.
“We have the CPA, a Rapid Diagnostic Tool for detection of Intestinal Bilharzia and also we have OMEGA 1, which is also a new tool which detect Intestinal Bilharzia. At the moment, we are running serum separated from whole blood. We collect whole blood from the human and then we test it with Rapid Diagnostic Kits, to check the presence of the antigens,” said Professor Midzi.
He added that the Rapid Diagnostic Tests were a simplified way of diagnosing Bilharzia or Schistomisias at the community level.
“At the moment, because we are developing the tool, we are still using blood. We will go from blood to urine. Why do we need to go from blood to urine? This is because urine can be produced willy-nilly without pain, but when you take blood, you have to insert a needle and take blood and taking blood is not pleasant so to increase compliance of the community in the surveillance of the disease, we use those tools that require the samples that are easy to be submitted by patients. We shall move from the serum, from blood to urine which is user friendly or which is field applicable.”
The pilot study that was launched in Zimbabwe is part of the Memorandum of Understanding (MoU) between the Ministry of Health and Child Care (MoHCC) and the China National Institute of Parasitic Diseases (NIPD). The study was meant to assess the various tools that will be used for the control of Vector Snails or the intermediate host snails that are responsible for the transmission of Schistosomiasis. The study was also assessing the Rapid Diagnostic Tests that can be used at a village or community level by the health facility facilities to improve surveillance of Bilharzia or Schistosomiasis at Primary Healthcare or community level where the diseases exist.

Professor Midzi said the development of Rapid Diagnoses Tests will help in the surveillance and early warning for rapid response for the control and elimination of Schistosomiasis.