Michael Gwarisa
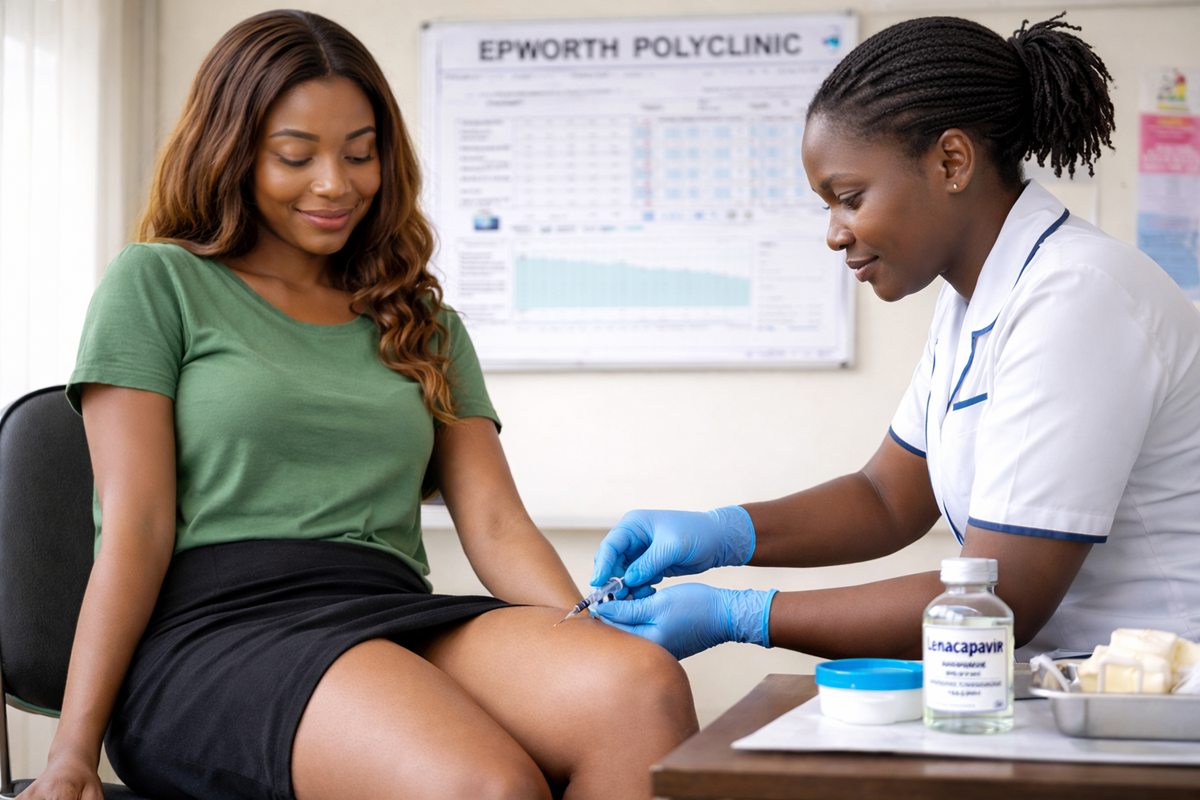
Global efforts to scale up access to a groundbreaking HIV prevention injection have received a major boost after PEPFAR and The Global Fund announced additional investment in Gilead Sciences’ long-acting drug, lenacapavir, with the aim of expanding protection to millions at risk.
The new commitment will support access for up to an additional one million people, bringing the total number targeted to three million across high-incidence, resource-limited countries by 2028. The move is expected to accelerate prevention efforts at a time when the global HIV response faces funding pressures and persistent new infections.
Lenacapavir, administered as a twice-yearly injection, is being hailed as a potential game changer in HIV prevention due to its long-acting nature and high efficacy. Unlike daily oral pre-exposure prophylaxis, the injectable option reduces adherence challenges that have long undermined prevention efforts.
“Lenacapavir represents one of the most important breakthroughs ever seen in HIV prevention, and Gilead’s priority is to ensure broad access as quickly as possible,” said Daniel O’Day, Chairman and Chief Executive Officer of Gilead Sciences.
“We expect large-scale generic rollout starting in 2027 and, in the meantime, Gilead is providing lenacapavir at no profit to the Global Fund and PEPFAR in countries with the most urgent need today. This additional commitment further expands access to lenacapavir as we build toward broad, affordable access and global impact,” O’Day added.
The collaboration between PEPFAR and the Global Fund is seen as critical in ensuring the drug reaches communities most in need. Both organisations have longstanding infrastructure in HIV programme delivery, particularly in sub-Saharan Africa, where the burden of new infections remains highest.
Lenacapavir is already making history as the first HIV prevention therapy to reach communities in sub-Saharan Africa in the same year it was approved in the United States, with initial shipments arriving at the end of 2025.
Globally, about 1.3 million new HIV infections occur each year, underscoring the urgent need for more effective and accessible prevention options. Long-acting injectable therapies like lenacapavir are increasingly viewed as essential to bending the curve of the epidemic.
There is currently no cure for HIV or AIDS.
Gilead’s broader strategy to expand access includes voluntary licensing agreements with six generic manufacturers, aimed at ensuring affordability and sustainability in low and middle-income countries. The company says it has accelerated steps to support generic production, with large-scale rollout expected in 2027.
Several countries and programme implementers have already begun engaging generic manufacturers in preparation for wider availability.
Lenacapavir works differently from existing HIV drugs. While most antivirals target a single stage of the virus lifecycle, lenacapavir is designed to inhibit HIV at multiple stages, reducing the risk of resistance and improving effectiveness. Laboratory studies have shown no known cross resistance with other drug classes.
The drug is also approved in multiple countries for treating multi-drug-resistant HIV when used in combination with other antiretrovirals, further highlighting its versatility.
In the United States, lenacapavir is marketed as Yeztugo for pre-exposure prophylaxis. It is indicated for adults and adolescents weighing at least 35 kilograms who are at risk of acquiring HIV. However, strict testing protocols are required to ensure individuals are HIV-negative before starting the injection.
Health experts caution that while lenacapavir offers strong protection, it must be used as part of a comprehensive prevention strategy that includes regular HIV testing, safer sex practices and continued risk assessment.
There are also concerns about the potential development of drug resistance if the injection is used by individuals with undiagnosed HIV infection or if dosing schedules are not followed correctly. Residual drug levels can remain in the body for up to a year after the last injection, making adherence to follow-up care critical.
Despite these considerations, the promise of a twice-yearly injection is generating optimism, particularly in regions where adherence to daily pills has been a challenge due to stigma, access barriers or lifestyle factors.
Zimbabwe, meanwhile, is preparing to integrate lenacapavir into its prevention strategy through a phased rollout targeting more than 40,000 people. Health officials say the initial focus will be on populations at highest risk of HIV infection, with gradual expansion planned as supply and delivery systems strengthen.
Public health experts say targeted rollout strategies such as Zimbabwe’s could play a crucial role in maximising impact, particularly in countries that have made progress but still face persistent transmission among key populations.
For Zimbabwe, the introduction of lenacapavir comes at a pivotal moment. The country has made significant gains in reducing HIV infections over the past decade but continues to face gaps in prevention, especially among young people and marginalised groups.
The phased introduction is expected to serve as a foundation for broader expansion as more doses become available and as health systems adapt to delivering long-acting injectable prevention.
As global partners scale up investment and coordination, stakeholders say the success of lenacapavir will ultimately depend on how quickly and equitably it can reach those who need it most.
If effectively deployed, the injectable could mark a turning point in HIV prevention efforts, offering a new tool to reduce infections and move closer to ending the epidemic.